Enveloped viruses have been detected in untreated wastewater, but scientists still don't fully understand the fate and infectivity of these viruses during water purification at treatment plants. Now, researchers reporting in American Chemical Society's Environmental Science & Technology have discovered that a standard water treatment technique, iron(III) coagulation, and its electrically driven counterpart, iron(0) electrocoagulation, can efficiently remove and inactivate a model enveloped virus.
Enveloped viruses have an outer coating of lipids and proteins that helps protect their genetic material. Typically, disrupting this coat inactivates the virus. Until now, most studies have investigated only disinfection by chlorine or ultraviolet light as a means to control enveloped viruses in wastewater. However, particles suspended in the water can sometimes shield viruses from disinfectants. Shankar Chellam and colleagues wondered whether coagulation with iron(III) can remove and inactivate enveloped viruses. They also wanted to study a related technique, iron(0) electrocoagulation, that shows promise for small-scale water treatment. As a model enveloped virus, the researchers chose an RNA virus, called Φ6, that infects bacteria.
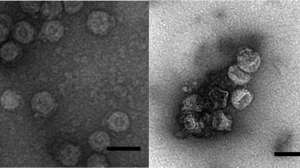
The researchers treated a solution containing Φ6 with either iron(III) or with iron(0) electrocoagulation, both of which formed iron precipitates. The hydrophobic viral envelopes stuck to the precipitates, allowing Φ6 to be easily removed as the solids settled. The conventional coagulation reduced the amount of active virus in the water by more than 100,000 times in 2.6 minutes, whereas electrocoagulation was slower but about 10 times more effective. The researchers observed that the structures of most of the viral particles in the iron precipitates were damaged, which rendered them unable to infect their host bacteria. Electrocoagulation also oxidized lipids in the viral envelope, further inactivating Φ6.
These results suggest that water treatment plants are already well equipped to remove enveloped viruses from drinking water by iron(III) coagulation, and viral levels are likely even further reduced by the additional treatment steps of filtration and disinfection, the researchers say.