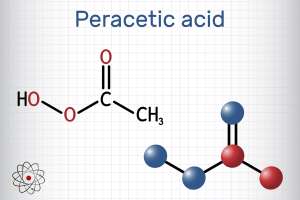
Chlorine and chlorine compounds, like sodium hypochlorite, continue to be the go-to chemicals for disinfection of water and wastewater. But the use of chlorine does have some drawbacks. For example, chlorine reacts with organic matter to form disinfection byproducts, and chlorinated water entering the environment may be harmful to plants, insects and animals. This has led to a growing interest in the use of peracetic acid which is highly effective against bacteria and other contaminants, does not form any known DBPs and has less environmental impact.
This content is sponsored by
Blue-White Industries.
Sponsored content is authorized by the client and does not necessarily reflect the views of COLE Publishing.
View our privacy policy.