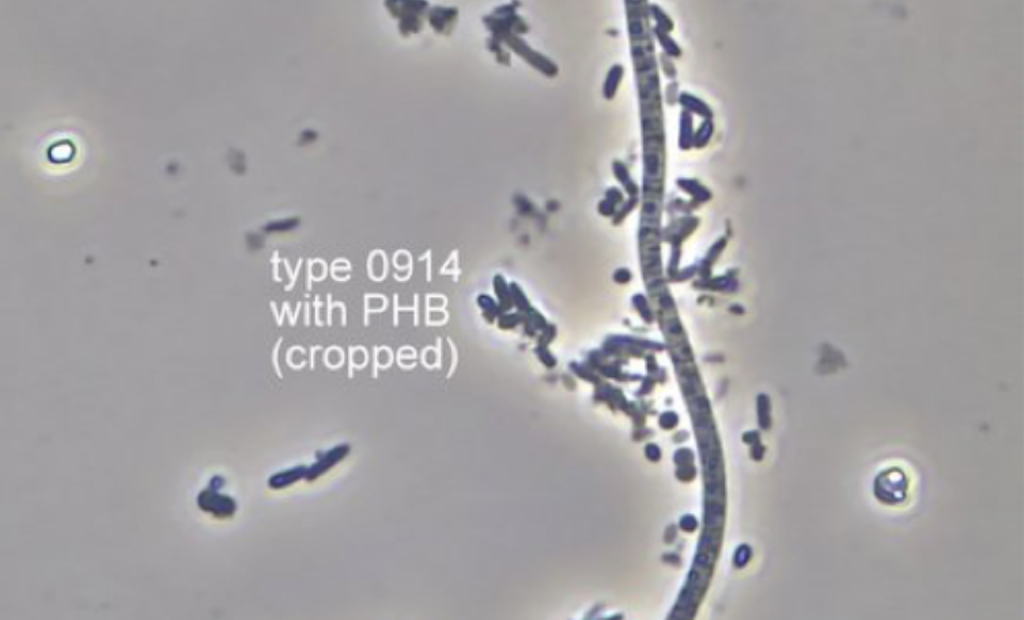
Filamentous bacteria Eickelboom morphotype 0914 is one of the most common filament types observed in all activated sludge treatment processes. Morphological features to recognize this filament are a cell diameter of approximately 1 µm, and squarish cell shape.
These filament...