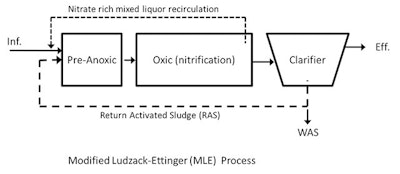
The Lab Detective was asked to provide some informal training about nitrogen removal in wastewater treatment plants, including a discussion of the various flow schemes used to remove nitrogen from the waste stream and return it to the atmosphere.
Always one to oblige, the...