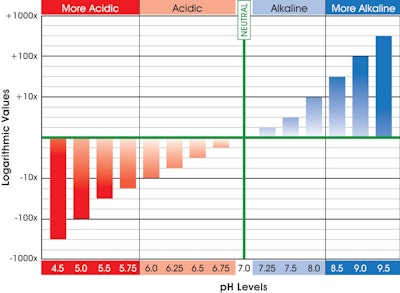
When operators are asked to define pH, many struggle to describe what it is and how it is measured. Let’s look at those matters — and explore how questions about pH might be worded on a wastewater licensing exam.
The technical definition of pH is: the logarithm of the...