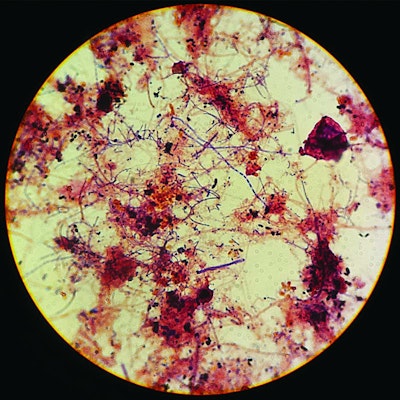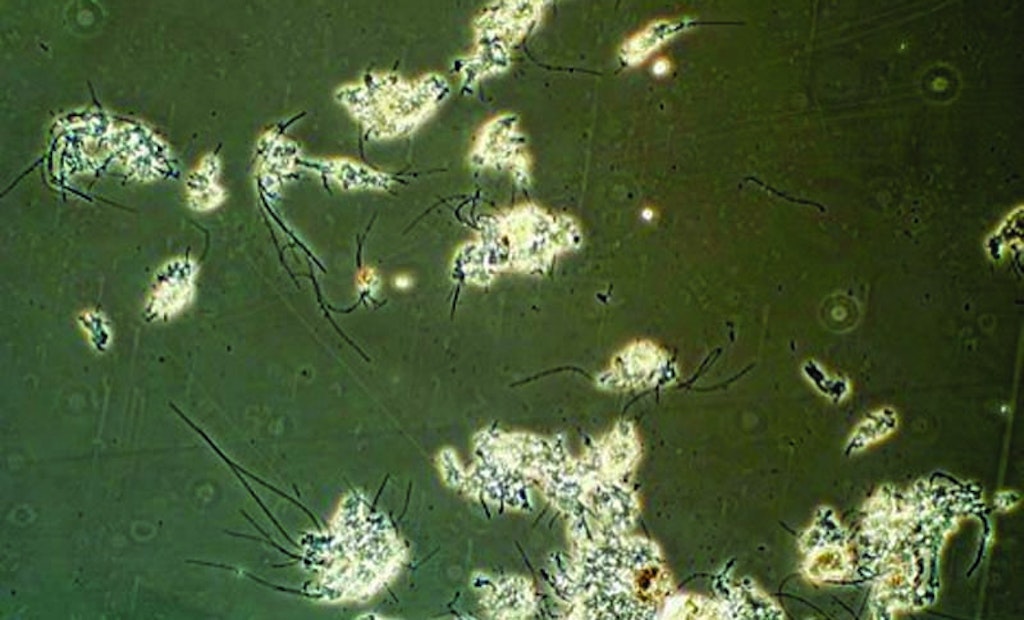
I have seen many changes during my wastewater career, now extending beyond 30 years.
SCADA systems and associated instrumentation have really taken hold in many public and private treatment works, enabling operators to monitor processes, open and close valves, adjust flow...